|
By Elizabeth Bach Ecosystem Restoration Scientist It’s a chilly, rainy spring afternoon, and I sit in front of the computer. Yet I can feel the heat of a sticky August afternoon, hear the whine of cicadas, and see the golden blooms of sunflowers. Mentally, I’m systematically walking through the prairie, carefully identifying all the plants. At Nachusa, many of us, myself included, find working outside in the prairies, savannas, and wetlands most rewarding. However, there is an incredibly important part of conservation work that happens at the computer: data entry and analysis. As the staff scientist at Nachusa, one of my primary duties is to analyze and share data. My primary tool for this work is a free program called “R.” In R, I can manipulate data, produce graphs, run statistical tests, and even produce a final report. Analyzing these data helps everyone at Nachusa refine restoration practices, inspires new ideas, and deepens our knowledge of the habitats and the organisms that live there. Sharing these data in presentations and publications allows us to share lessons learned and best practices used at Nachusa with others in both the conservation and scientific communities. In turn, we also learn from data from other sites. At Nachusa we are lucky to have several scientific researchers working at the site, who collect, analyze, and share data with us. We also have some data, collected over the years by The Nature Conservancy staff and collaborators, which haven’t been analyzed and shared. A key goal for Nachusa is to analyze these legacy datasets and share them. All this brings me back to my computer on an early spring afternoon. When there is less work to be done outside, I’m busy working with datasets on the computer, building graphs, thinking through which metrics best represent the observations made on the prairie, and building statistical models to understand how the Nachusa ecosystem has changed and how it might continue to change into the future. All this work is done with a few lines of code on the computer. While very different from the outdoor joys and challenges of data collection, there are both joys and challenges with this work. I often think of data analysis as a mystery to solve. What will the data show? What will I learn? How might this challenge or confirm observations from other scientists in other places? Every dataset is a new adventure, and I find a sort of excitement in that. It can also be frustrating. I spend a lot of time finding and correcting mistakes. There is no travel guide to inform my decisions. Fortunately, I can work with collaborators as travel buddies on these adventures, to bounce ideas off them, and gain a new perspective. One of the joys of working at Nachusa is being at the intersection of many paths of scientific research and natural history observation. Working with people with different expertise, skills, and perspectives deepens my understanding of science, the tallgrass prairie, and Nachusa. Elizabeth Bach is the Ecosystem Restoration Scientist at Nachusa Grasslands. She works with scientists, land managers, and stewards to holistically investigate questions about tallgrass prairie restoration ecology.
1 Comment
By Mary Meier Nachusa Grasslands volunteer Each May, Nachusa Grasslands’ staff and stewards usually dread the appearance of one of our major weed adversaries, reed canary grass (Phalaris arundinacea — RCG). This year, however, we may welcome the opportunity to attack the invaders, if and when we are released from our “stay at home” restrictions. The prospect of heading out into the field laden with herbicide backpacks is very appealing right now. What is reed canary grass? RCG is a coarse, cool-season perennial grass with erect hairless stems that grow from 2 to 6 feet tall. Densely clustered single flowers at the top of each plant change from green to purple to tan in late spring. Shiny dark brown seeds form during the summer months and shatter easily. Reproduction takes place both by seed dispersal and underground rhizomatous roots that create a thick, impenetrable mat just under the soil. Seeds can float down waterways and also spread via animals, humans, or machines. For example, Nachusa’s bison and deer populations may brush up against the plants and then carry the seeds in their fur. Where does reed canary grass grow? The plants thrive in moist areas, including marshes, swamps, prairies, meadows, fens, stream banks, and swales. It is especially abundant in disturbed wetlands, but can also appear in high quality native habitat. How did reed canary grass arrive in northern Illinois? Since the 1800s, agronomists have encouraged planting RCG for forage and erosion control. Some states prohibit selling the seeds, but Illinois does not. A native species actually exists, but it is almost impossible to distinguish from the more aggressive Eurasian variety. At Nachusa Grasslands we strive to eradicate all occurrences of RCG in order to diminish its ecological threats. Why does reed canary grass cause problems in our natural areas? RCG forms large monocultures, crowding out native species and building up a tremendous seed bank that germinates year after year. The thick thatch that forms from rhizomes and collapsed stems is especially problematic, as it prevents more desirable seeds from germinating. RCG, therefore, reduces native plant and insect diversity, while providing little shelter or food for wildlife. How do we manage reed canary grass at Nachusa Grasslands? Spraying grass herbicide is our main approach. The staff and stewards treat RCG with Intensity, a post-emergence grass herbicide (1% clethodim). Even though clethodim does not kill the plants’ roots, it helps set back the grasses and allows sedges and forbs to move in. Around waterways and high-quality natural areas, the crew uses the same formula with extra caution to reduce overspraying. What are some other reed canary grass control methods? Research and experience show that burning and mowing can actually stimulate regrowth of RCG but may also be useful in removing thatch prior to overseeding. Digging up rhizomes is labor-intensive and disturbs the soil, so other weeds may then invade the site. In small patches, cutting off the seed heads and disposing them off-site can be effective when combined with herbicide application. Covering with shade cloths is another option for large infestations. As with all weed management projects, best practices depend on overall goals and objectives, the size, distribution, and location of RCG infestations, willingness to use herbicides, and available human and equipment resources. In addition, every method requires follow-up monitoring, treatment, and establishing native species as we strive to extirpate this very challenging invasive species. Mary Meier has been a dedicated volunteer at Nachusa since 2002. She is currently an officer for Friends of Nachusa Grasslands, an Autumn on the Prairie festival organizer, and a member of the social media team. Along with her husband Al, she stewards the Dot and Doug Wade Prairie Unit, which is about half restoration and half remnant.
By Angie Burke Volunteer Coordinator, The Nature Conservancy We are all familiar with the saying “It’s the little things that matter”, and it’s the management of the tallgrass prairies at Nachusa Grasslands that has made a big difference for the littlest of things— mammals. Our paper “Early Small Mammal Responses to Bison Reintroduction and Prescribed Fire in Restored Tallgrass Prairies”, coauthored with Dr. Holly Jones and Dr. Nick Barber, sheds light on how the varying management of prescribed fire, coupled with the reintroduction of grazing bison, has created a habitat haven for the small mammals in a mix of agriculture and rural development.
blocking our safe access to a site, to capturing meadow jumping mice awakening from their winter slumber, every sampling season held a new adventure for us. Some of the little buddies we captured and released were deer mice, white-footed mice, prairie voles, northern short tailed shrew, meadow jumping mice, harvest mice, and my favorite, the 13-lined ground squirrel. Rain, snow, or shine, the little buddies are welcomed to the study each season with excitement by the many stewards, volunteers, and scientists that call Nachusa home. In the first two years since bison were reintroduced, we found fewer small mammals in older sites relative to new restorations and fewer as time since fire increased. Additionally, there was a higher diversity of what we did document in those older sites and slightly lower diversity (fewer than one species, on average) in sites where bison were present. This difference was driven mainly by prairie voles; fire removes litter and residual dead vegetation which is important habitat for voles. The overall abundance was especially influenced by the deer mice, which are able to use the areas with a higher prevalence of bare ground associated with frequent/recent fire on the landscape. Overall we found that bison reintroduction had fairly weak impacts to small mammal communities in the first few years. Bison, when reintroduced at a relatively low stocking rate, are not likely to cause significant shifts to this community or, by extension, to the seed predation and dispersal functions they serve in prairies. The many different types of habitat created by the managers at Nachusa varying prescribed fire with grazing bison maintain the diversity of small mammals on the landscape scale. Continuing to document the changes in the small mammals through time, while capturing the changes with other animal, invertebrate, and plant composition, will help to show how the little things matter on a big scale when it comes to tallgrass prairie restoration.
By Dee Hudson On March 7th Nachusa hosted a refresher day for the Nachusa and Middle Rock Conservation Partners fire crews. The day was sunny, and spring was in the air—a perfect day to light some prairie fires. How do you become a fire crewmember? The Nature Conservancy has three requirements for a potential fire crewmember: 1. Pass the pack test. This requires each person to carry a 24-pound pack for two miles. The trick is that the pack test must be completed in 30 minutes or less. Long-legged individuals have a bit of an advantage, while short-legged people really need to hustle. 2. Complete the S-130/S-190/I-100 online course work. These courses are self-paced and give an introduction to the basics of wildland fire training. There are questions at the end of each video section, and they must be answered correctly in order to continue to the next section. Before beginning the online course, candidates must contact Nachusa or another agency that will supervise the fire training, as a sponsor is needed to complete the online course. After the online portion is complete, there is one day of hands-on training. 3. Attend a fire refresher. Nachusa’s refresher this year focused on learning to use the pumper equipment and hand tools during live fire exercises. For more information about prescribed fire training, visit the Illinois Prescribed Fire Council website. To learn more about Nachusa’s fire program, visit the Controlled Burns page on the Friends of Nachusa Grasslands website.
By Jess Fliginger At first glance, it’s easy to mistake a fritillary butterfly for the well-known monarch; both can be seen fluttering across the prairie during the summer months, are similarly-sized, and are orange with black markings. In 2017 I spent the summer surveying for butterflies, particularly regal fritillaries and monarchs, in remnant prairies across the Loess Hills of Iowa. From my experience, the only way to get close enough to identify, or be fortunate to snap a photo of a butterfly is to move slowly and cautiously towards it as it’s fixed atop a flower, busily sipping nectar. Be prepared to pursue a fidgety butterfly for several yards as it swiftly drifts from one flower to the next. It took a great deal of practice and patience before I was able to become a stealthy butterfly ninja. Upon closer observation, the difference between a monarch and a fritillary butterfly becomes more apparent. There are 14 species of greater fritillaries (genus Speyeria) and 16 species of lesser fritillaries (genus Boloria). Both have a widespread range and can be found across the northern half of the United States into Canada, in some southern states, and parts of Mexico. Greater fritillaries inhabit woodland openings, meadows, prairies, and other open habitats where violets are present, while lesser fritillaries primarily live in wet meadows and bogs. Although greater fritillaries are much larger than lesser fritillaries, it can still be difficult to tell them apart while in flight. In total, there are 6 species of fritillaries that call Nachusa Grasslands home: great spangled fritillary (Speyeria cybele), regal fritillary (Speyeria idalia), aphrodite fritillary (Speyeria aphrodite), silver-bordered fritillary (Boloria selene), meadow fritillary (Boloria bellona), and variegated fritillary (Euptoieta claudia). The most common, and easiest to approach, is the great spangled fritillary. Of these, the regal fritillary is the only state-threatened species. A prairie-specialist species, regal fritillaries have drastically diminished throughout the Midwest, with only two localized populations remaining east of Illinois and several small isolated populations east of the Great Plains states and western Missouri. Typically, fritillaries have one brood and one flight period from June to August. Females lay their eggs near violets (Viola spp.), the caterpillar’s main food plant, in shady areas on the underside of dead vegetation. Soon after, the larvae hatch, crawl into nearby leaf litter, and sleep through the winter without feeding. During late winter to spring, the caterpillars begin munching on newly-sprouted violets and mature rapidly. Once fully grown, they pupate for several weeks until an adult fritillary butterfly emerges. Clearly, without violets there would be no fritillaries! Luckily for fritillaries, Nachusa has 7 species of violets throughout the preserve, as well as plenty of nectar sources to choose from. Equally as important to their survival, adult fritillaries require a large variety of nectar sources from native and non-native plants. I usually see them on coneflowers, goldenrods, ironweed, blazing-stars, milkweeds, mints, clovers, thistles – just to name a few. Plant any of these, along with violets, in your butterfly garden, and maybe a beautiful fritillary will pay you a visit. Jess Fliginger worked for Nachusa as a restoration technician during the summer of 2016. She has continued to be involved at the preserve, helping researchers conduct fieldwork and gather data. Working alongside Dr. Rich King in 2018 and 2019, she has collected data on Nachusa’s Blanding’s turtles . In addition, she has been volunteering with small mammal research since 2015, and worked for Dr. Holly Jones as a small mammal field technician in 2019. Lately, she has worked and volunteered in land restoration to enhance her skill set. She plans on assisting with prescribed burns at Nachusa this upcoming spring.
By Dee Hudson and Charles Larry Spring Summer Autumn Winter
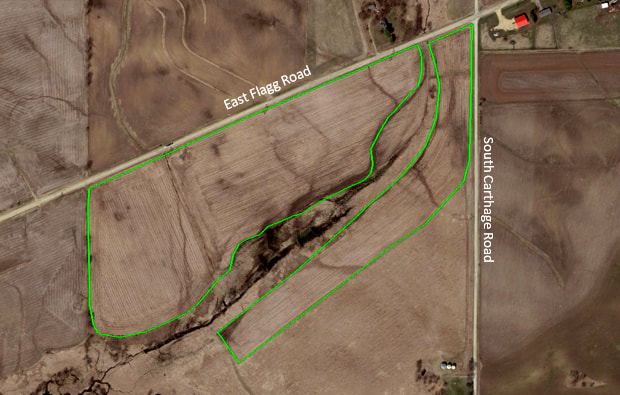
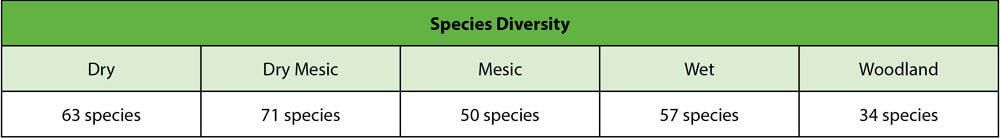
By Riley Nylin, Restoration Technician On November 18th, 2019 Riley Nylin, Tyler Pellegrini, and Amanda Contreras completed the 2019 crew planting on the corner of East Flagg Road and South Carthage Road. This 63-acre planting finishes off the Clear Creek Knolls management unit. Over the course of the season, our crew hand-picked 2,930 pounds of seed. Because of the extremely wet conditions of the picking season, we were forced to focus heavily on diversity instead of attempting to collect large amounts of seed. This led us to breaking only one seed collection record. We collected 29 pounds of pussytoes (Antennaria plantaginifolia) when the past record was only 14 pounds! Several different planting mixes were made, but not all of them were used on this site. The handpicked mixes are broken up into five categories: Dry, Dry Mesic, Mesic, Wet, and Woodland. The Wet and Woodland seed mixes were saved for other plantings/over-seeding areas. Within each mix, the crew focused heavily on species diversity. Table 1 displays the total number of species per mix. Once the seed was collected, separated, and mixed, the crew took to the field to plant! By planting 184 species at 50 lbs per acre, they planted a total of 85 acres of new prairie as well as over-seeding a few portions of past plantings. While 63 of the acres were planted at the Flagg and Carthage planting, the other 22 acres were planted at Franklin Creek Natural Area (FCNA). The FCNA planting was in partnership with the Illinois Department of Natural Resources.
By Jess Fliginger In 2013, Dr. Holly Jones started conducting a long-term research project at Nachusa Grasslands on quantifying the effects of disturbance-related management strategies on small mammal populations at restored and remnant prairie sites. The reintroduction of bison in 2014 allowed for a powerful before and after bison impact study that documented the effects of bison grazing on the small mammal communities. Data collected on species responses to bison, prescribed fire frequency, restoration age, and vegetation composition will inform decisions regarding abundance and biodiversity for small mammals. Small mammals play important roles in the food web by influencing vegetation structure through herbivory and seed predation, as well as serving as prey for predator species. So far, plant communities with bison grazing are becoming more diverse and more abundant with small mammals. In the beginning, Dr. Jones ran the small mammal project by herself for a year until she was able to pass it on to her Master’s student Angela Burke in 2014. It was quite a challenge to run the project on her own, and volunteers have become an essential component to keep it going. Over the years, we have had more than 100 volunteers participate to help check traps in the morning and reset traps in the afternoon. On the first day of small mammal trapping, or as we like to call it “smammaling”, we prep 150 metal Sherman traps by baiting them with peanut butter and oats. Our small army of volunteers, 3 or 4 people, create an assembly line, with one person spreading just a dab of peanut butter on the backplate and the other sprinkling a small pinch of oats inside. Once all traps have been prepped, we start stacking rows of them, Tetris style, in the back of Scarlet, our NIU mule.  Out of the four seasons we sample for small mammals, August and October have the tallest vegetation, making it difficult to locate our poles. We flag the highest plant we can find nearby; for me it’s usually prairie dock or good ol’ big bluestem, and we try to navigate our way through the meandering paths of the tallgrass prairie jungle." We take off to set 25 traps at six of our 5x5 grid sites, hoping our plans don’t get foiled by any bison delays or strange weather. Each site has flagged poles to indicate where the trap must be set; however, finding them can sometimes be a challenge. Bison love using our poles as backscratchers, and they are often found sprawled across the prairie. At each pole we place an open trap where it will sit until an unsuspecting critter passes by and catches a whiff of irresistible Jif.  The mice spend the night at their “mouse hotel” feasting on peanut butter and oats until we are back to process them in the morning. I always get a rush of excitement as I walk up to a trap and notice the door is closed. When I peek inside the trap, I am usually able to see a little face staring back at me. Occasionally, I’ll get a trigger-no-capture and my excitement will fade to dissatisfaction. Likewise, thieves are a constant problem. Some especially small, speedy daredevils are able to run in to the trap, take some quick bites of peanut butter, and run out without triggering it. We keep tabs on which traps have been thieved and adjust/replace them accordingly. To process the small mammals, we record the weight and take measurements on the right hind foot, tail, and body using a caliper. In addition, we determine their sex, age, reproductive status, species, and PIT tag number. Some of the species we have captured at our sites and record data on include thirteen-lined ground squirrel, deer mouse, white-footed mouse, western harvest mouse, meadow jumping mouse, prairie vole, meadow vole, and masked and short-tailed shrews. The most common species we capture is the deer mouse, Peromyscus maniculatus. Depending on whether it’s a new capture or recap, we will carefully insert a PIT tag underneath its skin – similar to microchipping your pet – as a way to keep track of its movements, survival, and reproduction throughout the study. It’s always a treat when we have an overwinter or recapture from the previous year; they were the lucky ones to survive the long cold winter! Finally, we provide complimentary haircuts to all new buddies and collect the hair to run in the stable isotope lab. The information gathered from each sample result can tell us about their diet and role in the food web. Since 2015, I have been volunteering with Dr. Jones’ small mammal project. This year I was given the opportunity to help run the project and process the small mammals until her incoming PhD student, Erin Rowland, arrived. I took up the challenge, and with practice I became a pro. I would say my favorite part of the job is meeting the volunteers and training them how to be great smammalers. I enjoy acting as a Nachusa tour guide to all newcomers, young and old. Although anyone is welcome to volunteer, the majority of our helpers are undergraduate students who enjoy a break away from the classroom. Volunteering for the small mammal project gets you to spend time outside, which is beneficial to your health and well-being. It inspires the public to engage in the scientific process, appreciate native plants and animals, and meet others who care about our environment. Furthermore, it helps develop team building skills that are important for any job setting. Volunteers are the heart and soul of the small mammal project, and without them I’m not sure it would be able to persist. There is a lot to accomplish within the 12 consecutive days we are at Nachusa smammaling, and any help is greatly appreciated! If you are interested in volunteering, please contact Erin Rowland. To me, the small mammal project is all about making new and old friends — volunteers and mice alike. Small mammal research has been supported by the Friends of Nachusa Grasslands science grants from 2015 to 2018.
Consider a donation to the Friends of Nachusa Grasslands to support the ongoing science! “Budding Ecologists” — Nachusa’s Role in Mentoring the Next Generation of Natural Areas Managers9/15/2019 By Cody Considine
In her new role she will lead the crew for the remainder of this field season, which includes harvesting a couple more thousand pounds of seed from 150 species to plant 50+ acres this fall. In addition to the fall planting and prescribed fire season, she will also be interwoven into the bison roundup, helping with various tasks. This winter she will operate chainsaws and large equipment, removing brush in our oak woodlands. Next spring she will work to become qualified as a line boss position on prescribed fire. By the end of her residency (December 2020), Amanda will have the skillsets, confidence, and humility to be a natural areas manager.
Bill and I, along with Elizabeth and Dee, would agree that one of the most gratifying experiences in managing natural areas is helping grow the next generation of natural areas managers. We are immensely grateful for all of our young professionals who choose to start their careers at Nachusa. Please give them a shout out next time you come for a visit. Cody Considine is the Deputy Director at The Nature Conservancy’s Nachusa Grasslands. By Jason Willand, PhD I first visited Nachusa Grasslands in August 2008 while I was working for the Illinois Natural History Survey. I was overwhelmed by the sheer scale of the restorations that comprised the preserve and never envisioned myself conducting research on these restored prairies. As fate would have it, I returned to school in 2009 to start work on my doctorate degree and was able to fit part of my research into the restorations at Nachusa. The research was for the first chapter of my dissertation, where I examined the role of seed and bud banks for plant community regeneration during prairie restoration. The field portion of this work lasted only five days, and afterwards I was hoping that I would have a chance to return to conduct more research. As fate would have it again, I was able to conduct a small research project at Nachusa as I was wrapping up my dissertation in July 2014. The research project was the result of brainstorming between my dissertation advisor Sara Baer and myself. With the imminent introduction of bison on the preserve in October 2014, we wanted to develop a potential long-term monitoring project. We decided that an interesting study would be to examine the resource availability of the remnant and restored prairies before the bison were introduced. Bison were the dominant grazers in the tallgrass prairie ecosystem before settlement by the pioneers. They play a “keystone” role in the maintenance and diversity of prairies because of their wallowing behavior and preferential grazing on graminoids (grasses and sedges). Most bison research to date has been conducted either on private game ranches or remnant prairies, with little research coming from restored prairies. We collected data on three resources that could affect where bison would graze in the introduction area: plant biomass, the forage quality of the biomass, and soil carbon and nitrogen. Knowledge of plant biomass provides a rough estimate of the amount of plant matter available for bison consumption. Forage quality of plant biomass is informative because it not only tells us how much of the plant matter is actually digestible to the bison, but also the fat and crude protein content of the plant matter. Soil carbon and nitrogen are vital because as a plant uptakes them, they allow a plant to produce important macromolecules for growth, such as proteins. In order to adequately sample the bison introduction area we surveyed three different prairie types: remnant prairies, restored prairies more than 15 years old and restored prairies less than 5 years old. To quantify potential differences in resource availability between the three prairie types we collected plant biomass and soil samples from three different “fields” in each prairie type. Both the plant biomass and soil samples were returned to the laboratory at Southern Illinois University, where they were processed. Forage quality samples were sent to the University of Wisconsin Madison Soil and Forage Laboratory for analysis of seven components of forage quality. We found that the restored prairies less than 5 years old had almost twice the amount of plant biomass compared to the restored prairies more than 15 years old and more than twice that of the remnant prairies. Surprisingly, there was little difference in forage quality and stored carbon and nitrogen in soil among the three prairie types. The similarity in forage quality between the three prairie types may be attributed to prescribed burning, as all the fields were burned in April 2014 three months before we sampled them. Prescribed burning has been found to increase forage quality for up to a year after a fire and may have created homogenous plant biomass on the landscape. We expected soil carbon and nitrogen to be higher in the remnant prairies because these soils have not been tilled, a disturbance that has been found to reduce the storage of carbon and nitrogen in agricultural soils. The remnant prairies we sampled perhaps had a lower storage of carbon and nitrogen than expected because the soil was fairly shallow in comparison to the typical deep, loamy soils that characterize many remnant prairies. The findings of this study suggest that bison may prefer the youngest restored prairies because there is simply more plant biomass available and little difference in the forage quality from the other prairie types. Even with these preliminary data it is still difficult to predict where bison will graze. Other factors that need to be considered are the dietary preferences of male and female bison and how prescribed burning creates a more heterogeneous landscape in the three prairie types. Post-introduction data have not been collected, so at this point any predictions of landscape use by bison is speculative at best. Maybe fate will strike again and I will be able to collect more data at Nachusa sometime in the near future. Jason Willand is an associate professor of biology at Missouri Southern State University in Joplin, MO where he currently serves as the assistant department chair and chair of the conservation section of the Missouri Academy of Sciences.
|
Blog CoordinatorDee Hudson
I am a nature photographer, a freelance graphic designer, and steward at Nachusa's Thelma Carpenter Prairie. I have taken photos for Nachusa since 2012. EditorJames Higby
I have been a high school French teacher, registered piano technician, and librarian. In retirement I am a volunteer historian at Lee County Historical and Genealogical Society. Categories
All
Archives
January 2024
|
CONNECT WITH US |
8772 S. Lowden Road (mailing address) Contact Us
|
|
© 2024 FRIENDS OF NACHUSA GRASSLANDS The content on this website is owned by us and our licensors. Do not copy any content (including images) without our consent.
|

































































 RSS Feed
RSS Feed