|
By Dee Hudson and Charles Larry SpringSummerAutumn Winter
3 Comments
By Kirk Hallowell and Mary Meier Nachusa Grasslands Volunteers Each year, volunteers commit over 8,000 hours of service to the restoration efforts of Nachusa Grasslands. Volunteers harvest seeds, keep invasive species at bay, assist in scientific research, tend prescribed fires, and perform a myriad of other tasks essential to the restoration process. Volunteer projects must be supported by equipment, herbicide, contract services, and other resources needed for success. Fortunately, the Illinois Clean Energy Community Foundation empowers volunteers by providing grant funds as a match to local dollars raised and labor donated. Community Stewardship Challenge Grant Program awards are used to purchase equipment, herbicide, and services necessary to make these volunteer activities possible. According to its website, “ The Illinois Clean Energy Community Foundation (ICECF) was established in December 1999 as an independent foundation with a $225 million endowment provided by Commonwealth Edison. Our mission is to improve energy efficiency, advance the development and use of renewable energy resources, and protect natural areas and wildlife habitat in communities all across Illinois.” (@illnoiscleanenergycommunityfoundation) The Illinois Community Foundation will award Friends of Nachusa Grasslands a grant of up to $30,000 if we fulfill requirements under several categories between May 1, 2023, and April 30, 2024:
Under the leadership of project coordinator, Kirk Hallowell, Friends has chosen two sites at which to focus volunteer hours and grant resources for their current stewardship project: Nachusa’s Big Jump Unit and the Holland Savanna Unit. Both areas are infested with invasive shrubs, including autumn olive (Elaeagnus umbellata) and amur honeysuckle (Lonicera maackii), two of the most tenacious foes that Nachusa’s volunteers battle. On its "Where We Work" web page, The Nature Conservancy explains, “Autumn olive is a problem because it outcompetes and displaces native plants. It does this by shading them out and by changing the chemistry of the soil around it, a process called allelopathy. Loss of native vegetation can have cascading effects throughout an ecosystem, and invasive species are one of the major drivers for a loss of biodiversity.” According to a Friends of Nachusa Grasslands blog post by Scientific Research Grant recipient Kaleb Baker, “Amur honeysuckle is an invasive shrub that flourishes along forest edges and in open woodlands such as those at Nachusa Grasslands. Amur honeysuckle shades out native flora with its early leaf-out and prolonged leaf retention, and when left uncontrolled, it can produce a near monoculture, threatening biodiversity.” Some of the ICECF funds already awarded have been invested in herbicide effective in eradicating autumn olive and amur honeysuckle. Volunteers treat the plants using backpacks and handheld sprayers. The next facet of the Big Jump project is to cut down and stack hundreds of non-native trees to make room for prairie restoration. This task that requires heavy equipment owned and operated by an external tree service contractor. ICECF grant funds will also be used to pay for the machinery and professional tree fellers. Kirk, who also serves as steward of the Holland Unit, offered, “It is exciting to know that the efforts of our volunteers empowered by the ICECF grant resources will make a lasting impact on our efforts to control amur honeysuckle in the Holland Savanna.” An additional challenge for the Holland Unit is that the parcel is dominated by cool weather grasses, including rye and fescue, which are typically resistant to overseeding efforts to establish native prairie plantings. In the second half of the ICECF grant period, we will experiment with preparation methods using herbicide to loosen the grip of the dominant grasses and allow overseeding of native plants to take hold. During the fall months, volunteers also helped collect seeds from the preserve for enhancing our prairie plantings. Our long-term goal is to establish a diverse prairie planting on both the Big Jump and Holland Savanna sites providing for long-term weed management and suppression of non-native shrubs and trees. Ongoing stewardship efforts, including volunteer labor, herbicide application, and controlled burns, will gradually help integrate the target areas into the surrounding habitat. The Friends Social Media Team uses Facebook, Twitter/X, Instagram, and our website to promote volunteer opportunities. You can also follow Illinois Clean Energy Community Foundation’s Community Stewardship Challenge Grant Program on Facebook and Twitter/X to learn more about the Foundation. #CSgrantsIL #NAicecfdn How Can You Help Friends Earn the ICECF Stewardship Grant?
Volunteer for a Thursday or Saturday brush clearing workday — check SignUpGenius for the schedule. Meet at Nachusa’s Headquarters Barn before 10 am and be ready to restore habitat. If you have any questions about the workdays, check the Friends of Nachusa Grasslands website. By Josh Klostermann Doctoral Student, Department of Plant Science and Technology at the University of Missouri Have you ever observed a bee tenaciously collect every grain of pollen out of an anther and then zoom off into nothingness? Where did that bee go? An easy answer to that question is back to its nest, but the nesting habits of most bees are mysterious. Where each different species decides to build a nest is poorly understood and not frequently observed. So, it may be more difficult to locate that pollen-loaded buzzer than originally expected. It is at the intersection of this problem and Nachusa Grasslands where my passion for a good hunt, infatuation with natural history observation, and the regionally uncommon bee Anthophora abrupta collide. But first I will give a little backstory. Most bees are solitary, and most bees that are solitary are soil excavators. In other words, a large majority of the bees on this planet live alone (without a hive or queen) and dig underground to build their nests. Where a solitary mother bee chooses to build her nest is arguably one of the most important decisions she will make in her life. There is an immense pressure on her to be able to pick a nesting site that will allow her to operate as efficiently as possible while also reducing the risk of nest failure through predation, parasitism, or infection from pathogens. Once committed to a nesting site she will invest all her energy on that place, visually and chemically remembering where it is, provisioning the cells she creates with hard foraged pollen, and laying eggs on those provisions to ensure that more of her kind will emerge the following year. With these responsibilities, it may be safe to assume that bees must be keen on detecting differences in above and below ground microhabitats in order to choose a site that will allow them to be as fecund as possible in their short lifetimes. Some examples of microhabitat differences that would be relevant to a nest founding bee may be the amount of bare ground at a site, the amount of sunlight the site receives, and the texture and compaction of the soil. The degree to which these differences in microhabitats filter what can exist in a community is largely unknown and an important puzzle piece in understanding how bee species persist. As a researcher, I am focused on how the presence of these microhabitats affect what bees and wasps nest where in a landscape and how disturbances alter the landscape to create many different types of microhabitats for bee and wasp nests. Disturbance is an unavoidable reality in ecology. It is often painted in bad light due to our human ability to disturb so profoundly that it alters communities indefinitely, but not all disturbance is bad. In fact, small scale disturbance at intermediate intervals has been shown to increase the diversity of organisms in an environment and promote co-existence. Therefore, identifying mechanisms of small-scale disturbance that provide this sort of stability to bee and wasp communities is essential to their conservation. One mechanism of disturbance I am becoming increasingly interested in is wind. When a mature tree is downed by wind, several different things happen at a microhabitat scale. First of all, the root system is uplifted and a pit of exposed ground with an associated vertical wall of a soil-root matrix is created. These soils are fundamentally different from the surrounding soil just by the nature of being covered and ensconced in tree roots for decades and then one day suddenly uplifted. Also, canopy cover decreases, and there is an influx of light into the immediate area adjacent to the downed tree. This creates temperature and moisture differences from the surrounding woodland. The disturbance of a freshly-downed tree creates an array of free “real estate” that a bee or wasp may find suitable as a nesting site. This real estate is a hot commodity, and downed mature trees are rare in today’s forests. As time moves on, more trees become downed, and the bees and wasps that utilize these spaces disperse to fresh sites. I believe that this sort of dynamic, small-scale, and intermediate disturbance may be a crucial component to the stability of bee and wasp populations that nest in woodlands. In the summer of 2021, I scoured the wooded areas of Nachusa grasslands and surrounding forests looking for these “tipped-up” microsites. By targeting these places, I have been able to confirm that they are indeed being utilized as nest sites for several different species of bees and wasps. An important discovery was finding Anthophora abrupta at three different sites in Northern Illinois all nesting in the soil-root matrix of “tip-ups”. This bumblebee look-alike is regionally uncommon and was a site record for Nachusa Grasslands. While carefully observing the nesting sites of A. abrupta I was also able to spot their associated cleptoparasite; Brachymelecta californica. A cleptoparasite is an organism that steals resources from another. B. californica will search for nests of A. abrupta, sneak inside, and lay its eggs on the pollen provisions collected by their host. This bee is extremely rare to the region and was a first record for the state of Illinois. Other notable members of the “tip up” community thus far are Myrmosa unicolor, Philanthus gibbosus, Pseudomethoca frigida, and small metallic sweat bees in the Lasioglossum subgenus Dialictus. There is so much more to bees and wasps than flowers and stinging. I hope that this blog post has opened a new window for looking at these small insects with curiosity and wonder. It is through careful observation and patience that insight may be passed from the non-human to the human. The actions of an invertebrate may seem insignificant, but these organisms have been surviving on earth for much longer than us. Next time you’re in the prairie in the sweltering humidity of midsummer and the droning of bees swerving between pink rays and into white corollas feels disorganized, even chaotic, take a swig of water, a bite of lunch, and a closer look: those supersonic meanders may begin to look like slow dances with each bee’s movements holding purpose.
Friends of Nachusa Grasslands awarded Josh Klostermann Scientific Research Grants in 2022 and 2023. Are you interested in supporting research projects at Nachusa? Just designate your Donation to "Scientific Research Grants."
By Dee Hudson and Charles Larry SpringSummerBy Marissa Gorsegner Nachusa Restoration Technician When you look around Nachusa Grasslands in the summer, you can see constant change in the prairie. You see an array of people day in and day out working in the summer heat. Some of these people are the summer restoration crew whose job is to work on the restorative vision of Nachusa. We spent our time working hard at killing weeds to restore the prairie, picking seeds for future plantings and, of course, having fun! Our 2022 summer restoration crew was made up of seven members who never said no to a challenge that presented itself. Working together, we faced these challenges head-on. One issue every summer is fighting the weeds that pop up throughout the prairie. We began early summer with focusing on reed canary and bird’s foot trefoil (BFT). Then we moved onto king devil. Yellow and white sweet clover became the next major focus, with BFT moving right back to the top of the list as seed pods began to pop up everywhere. We also busied ourselves with collecting seeds, collecting more and more as the summer progressed. Seeds such as pussytoes, oxalis, daisy fleabane, sedges, and June grass were some of the favorites to collect. We also learned how to mill seeds, and spent some time during rainy days milling seeds we had collected.
SUMMER CREW
FALL CREW Along with Molly Duncan and Derek Hofland, Noah and Booker joined the fall crew.
By Devin Edmonds Doctoral Student, Department of Natural Resources and Environmental Sciences University of Illinois at Urbana-Champaign Turtles are arguably the most liked of all the reptiles. Unlike their legless and slithery counterparts, people rarely fear turtles or worry about their bite. We keep turtles as pets, buy turtle-branded products, and associate turtles with good luck. Turtles are the one group of reptiles most people actually like. But aside from being likable, turtles also serve important ecological roles. Turtles help cycle nutrients from water to land, supporting energy flow between terrestrial and aquatic ecosystems. Some species are ecosystem engineers, reshaping their physical environment with burrows. Turtles have even been shown to play an important role in seed germination and dispersal. Despite their importance, many turtle species are in trouble. There are more than 300 different kinds of turtles and tortoises in the world; over half are threatened with extinction. Of our 17 Illinois turtle species, the Department of Natural Resources lists six as Endangered and one as Threatened. World Turtle Day was established in 2000 to bring attention to the precarious situation many turtles face. Nachusa Grasslands provides important habitat for five turtle species: Midland painted turtle (Chrysemys picta marginata) The Midland painted turtle is the most likely turtle to be seen at Nachusa Grasslands. They are aquatic but often can be observed on sunny days basking on logs or other objects protruding from the water’s surface. Their most easily identifiable feature is their yellow-striped head. Painted turtle populations are stable in Illinois, and the species is common and widespread. They are more adaptable to habitat degradation than other turtles in the state. Common snapping turtle (Chelydra serpentina) The largest turtle at Nachusa Grasslands is the common snapping turtle. These monsters are almost entirely aquatic and go unnoticed for most of the year. In June, females emerge from the murky depths to lay eggs on land, and it is during the late spring-early summer nesting season when you are likely to encounter one. Though they look menacing, in the water snappers are quite shy and reclusive animals. Snapping turtles are adaptable, widespread, and found throughout most Illinois waterbodies. Eastern spiny softshell turtle (Apalone spinifera spinifera) The most peculiar-looking turtle at Nachusa Grasslands is the Eastern spiny softshell. Their shell is not hard, but rubbery in texture, and their feet are fully webbed. Females are twice the size of males, growing up to around 15 inches in length. When softshells feel threatened, they dive underwater and burrow below the substrate with only their head sticking up from the muck. They tend to be associated with lakes, rivers, and larger bodies of water. At Nachusa, spiny softshells have been seen in Clear Creek but may occur elsewhere in the preserve. Ornate box turtle (Terrapene ornata) The ornate box turtle is the only land turtle at Nachusa Grasslands. They are a true prairie species, restricted to sand prairies and the southern till plain of Illinois. Since much of their habitat is now agricultural land, Illinois populations are small and isolated. Consequently, the ornate box turtle is state listed as Threatened. They are easy to identify at Nachusa by the attractive radiating pattern of yellow or cream-colored lines on their shell. For over a decade, Nachusa Grasslands has been supporting research on the species, which is helping inform conservation efforts. Blanding’s turtle (Emydoidea blandingii) The most threatened turtle species found at Nachusa Grasslands is Blanding’s turtle. Their most prominent feature is their bright yellow chin and throat. They are semi-aquatic and restricted to prairie wetlands and wet meadows, where the water is calm and there is ample vegetation. They make long treks overland to nest and occasionally travel between wetlands. Habitat loss is the main threat to Blanding’s turtles, with Nachusa Grasslands providing critical habitat for this state Endangered species. On top of habitat management, Nachusa has also helped initiate a head-starting program for Blanding’s turtles, whereby eggs are removed from nests, incubated in captivity, and baby turtles released after being raised to a size where they are less vulnerable to predation. So far, more than 70 one year-old Blanding’s turtles have been released in native prairie habitat as part of the head-starting program. To help turtles, donate to:
By Charles Larry Friends of Nachusa Grasslands Board It was the Greek philosopher Empedocles (c.450 BCE) who first presented the idea of the four elements: earth, air, fire, and water. He did not call them elements, but roots, and conceived of these not in terms of the physical properties we associate with the words, but as forms of energies whose various combinations make possible the structure of the physical world. This idea was later amplified by Plato, and then, especially, Aristotle. For the purpose of this blog post, however, earth, air, fire, and water are being used more as a conceit to imaginatively explore the elements as they may (loosely) apply to Nachusa. This exploration, in part, will utilize stories from Greek mythology. EarthHeavy, dense, solid: earth. As Gaia the earth is the foundational mother of all living things. The name Gaia is now associated with many environmental concerns. Is there any North American mammal more evocative of earth than a bison? Massive, strong, brown in color, the bison seems sculpted of earth. Rock: the bones of the earth. Nachusa's rock formations are mainly St. Peter Sandstone, which is composed of sand that once rested on the bottom of ancient seas. Over billions of years this sand sediment compacted and eroded, and after extensive land mass shifts with tectonic and glacial activity, the rock became exposed and left as outcrop. The earth with its layer of soil, giving foundation and nutrients and collecting moisture, provides growing things the necessities of life. The earth establishes a stable environment for plants, animals, and all creatures of the air. AirLight, open, space: air. Air has been called the breath of life. It surrounds the planet in a protective layer and is a requirement for most living things. In The Odyssey, Aeolus, keeper of the winds, put all of the winds except the west wind in a bag and gave it to Odysseus. The west wind would take Odysseus and his men directly to their island home of Ithaca. But the men, thinking it was some treasure that Odysseus was keeping for himself, opened the bag and let loose the winds which drove them far from home, thus taking an additional ten years to reach home after having fought in Troy for ten years. There are times at Nachusa when it seems that bag has been opened on the prairie. The early pioneers, traveling across the tall grass prairie and observing the undulating waves of grasses caused by the wind, called it a "sea of grass." Wind also helps disperse seeds, such as the lithe milkweed seed. Mankind, observing birds and flying insects, dreamed of taking to the air long before it became a reality. Daedalus, who designed the labyrinth imprisoning the Minotaur, himself became imprisoned, along with his son Icarus, to keep the secret of the labyrinth. Daedalus crafted wings made of wax so that he and Icarus could escape. This succeeded until Icarus, in his youthful headiness, flew too close to the sun, melting the wings, thereby plunging him into the sea, resulting in his death. There are times when the air, the sky with its cloud formations seems to be on fire. FireHot, expansive, light: fire. Fire can be both constructive and destructive. In Greek mythology, Prometheus stole fire from the gods and gave it to mankind as a gift, thereby creating technology and civilization. For this act he was punished by the gods by being chained to a rock, where an eagle each day ate his liver, which grew back overnight, only to be eaten again, over and over. At Nachusa, fire usually means a prescribed burn. The prairie, like the Phoenix, needs fire for renewal. Our most pervasive source of fire/light is the sun. Sunlight is needed for plants to photosynthesize. Illuminating everything, the sun can seemingly perform magical transformations, such as becoming like molten gold on water. WaterCold, contractive, fluid: water. Most of our planet is covered by water. Perhaps the most versatile of the elements, water can exist in different states. As a fluid it is water, as a gas it is steam or mist, and as a crystalline solid it is ice. In Greek mythology it is Poseidon who rules the waters. Better known by his Roman name: Neptune, he is the Old Man of the Sea, covered by scales and holding a trident. Poseidon is the protector of all who live in the waters of the earth. It is Nachusa's ponds and streams which provide the life-giving element to all the creatures at home here. The most common source of water at Nachusa is, of course, rain. Life-nourishing rain sweeps across the prairie and savanna in storms. In winter, snow often blankets the land with needed moisture. Water: the lifeblood of the earth. If you would like to volunteer at Nachusa Grasslands, consider joining our Thursday or Saturday Workdays.
By Dee Hudson and Charles Larry SpringSummerAutumnWinterBy Luke D. Fannin PhD Candidate, Dartmouth College, Hanover, NH Grass-eating, or graminivory, as it is called by scientists, is a strange behavior. At the outset, grasses, at least compared to the many plant foods that humans consume regularly, look unappetizing; they are tough to chew, they are full of fibers that make them difficult to digest, and they are often covered in dust and sand from growing close to the soil surface. But for many mammals, ranging in size from tiny voles (20-24 grams) all the way up to gigantic white rhinoceroses (2400 kilograms), grasses are dietary staples, and these so-called “grazers” (grass-eating mammals) are pivotal in Earth ecosystems. Nachusa Grasslands is home to one of North America’s most important grazing mammals, the bison (Bison bison), which eats grasses in most months of the year. But if grasses are such difficult foods to eat, how do bison–let alone any other mammals–eat them? As it turns out, bison have a few tricks up their proverbial sleeves as it pertains to eating grasses. For starters, bison are ruminant mammals, which means they have highly specialized stomachs that allow them the ability to regurgitate and then re-chew partially digested plant foods. Think of how a domestic cow eats; the cow first swallows a bite of food but then proceeds to regurgitate that bite of food and chew it again, and again, and again… until finally those food particles are small enough to pass through the rest of the digestive system. With each subsequent swallow, foods are bathed in stomach juices teeming with bacteria, which also help to weaken the structural integrity of fibrous foods and liberate nutrients. This digestive trick is incredibly helpful for bison, as it allows them to digest grasses in a way that humans cannot. Secondly, bison are endowed with highly specialized teeth, termed hypsodonty. Unlike the surfaces of our own teeth, which fully poke out of our gums when they erupt, bison’s teeth emerge in a piecemeal fashion over the course of their lives, meaning that they usually have more teeth hiding within their skulls at any given point in time. A good analogy for how bison teeth work is the lead found in the tip of a mechanical pencil: while there is always lead at the pencil tip exposed for writing, there is far more lead stored within the pencil that emerges to eventually replace the used-up lead after writing is finished. Hypsodonty is thus a useful trait for Nachusa bison because most grass plants are also abrasive (see below), meaning they remove tooth surface, or enamel, during chewing. Hypsodonty allows bison to continuously provide new replacement tooth surface over the course of their lives, allowing them to keep eating grass plants without having to worry about completely losing their teeth in the process (although they are not actively growing teeth, and this excess amount of tooth eventually wears out in old age). 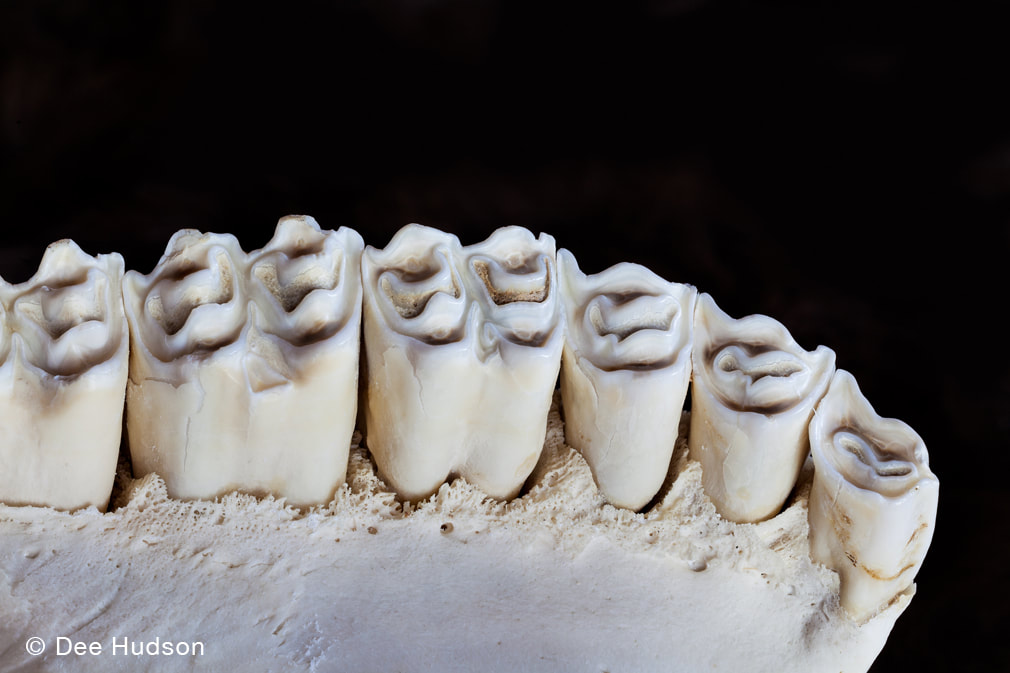 Bison teeth are hypsodont, meaning that the dental crowns are tall and most of this length is stored within the bones of the face and jaw. This trait provides a hidden reservoir of tooth material that can be pushed out when the surfaces of the teeth wear, allowing for tooth function to be preserved in the face of extremely abrasive conditions. In addition, when bison teeth wear, the enamel forms elaborate cutting surfaces with a harder underlying tooth material (referred to as dentin, which is the brown material contrasted against the pearly-white enamel on the tooth surfaces). These elaborate cutting surfaces help bison mince plant foods. But with all this talk of bison traits to eating grass, it is easy to view the grasses themselves as passive players in this story of herbivory at Nachusa. But can grasses fight back? My own work at Nachusa Grasslands seeks to answer this question with one plant trait that may act as a defense against herbivory . . . silica levels. While we often think of silica as being a component of our modern technology (e.g., in computer chips), silica is also one of the most abundant minerals in Earth’s soils. Grasses, in turn, uptake silica into their own tissues during growth, where it usually gets deposited into silica bodies called phytoliths. The function of silica accumulation in grasses is debated, but one thought is that it provides a defense against defoliation (i.e., the removal of leaves). As suggested previously, grass plants are abrasive, and it is the silica in grass leaves that hypothetically works to remove enamel. As such, high levels of silica in grass leaves may deter mammalian consumers from eating them — for risk of damaging their teeth — helping to prevent defoliation. Silica also works to stiffen plant leaves, making them more difficult to chew and digest, while also further inhibiting microbial digestion in the digestive tract. Thus, silica be an inducible defense for grasses, wherein a re-growing grass plant responds to previous herbivory by increasing silica uptake to prevent severe defoliation in the future. But silica has other important functions in grass plants that are unrelated to herbivory. For example, increased silica uptake in grasses is also important for relieving environment stresses related to both high temperatures and low water availability. Therefore, it remains challenging to determine whether the high silica levels found in some prairie grass plants are primarily a response to intense herbivory pressure (i.e., from roaming bison) or are instead a plastic response related to changes in local environmental conditions that may increase growing stresses.  Bison at Nachusa can exhibit immense herbivory pressure on the local grassland community and repeat herbivory at certain locations can create what are termed “grazing lawns”, where grass plants are maintained in a short-statured state as compared to surrounding vegetation. It remains unknown whether the grass species within these “grazing lawns” at Nachusa possess higher silica levels in their tissues than similar species within exclusion plots, but previous work on the African Serengeti has suggested that grasses in such features are more silica-rich than similar species under less extreme herbivore pressure. My current work at Nachusa Grasslands seeks to shed light on this mystery. By taking advantage of bison exclusion plots set up across the lands of the reserve, I can compare the silica levels of different grass species (and other related traits, such as photosynthetic pathways, leaf toughness, and nutrient levels) in locations where they have experienced almost a decade of bison herbivory (2014 – 2021) to those same species in exclusion areas where bison herbivory has been much less intense or non-existent. My hope for my continuing work at Nachusa is to disentangle the various factors that may influence silica levels in various grass tissues of prairie plants and, by extension, in the diets of Nachusa bison. My overarching goal is to figure out if re-introduced bison are helping to change the functional traits of grasses growing within the Nachusa reserve, and if so, what this might mean for plant community structure and resilience moving forward.  Exclosure or herbivore exclusion plots are key to testing hypotheses regarding inducible plant defenses and bison herbivory at Nachusa Grasslands. The bison have access to the plants on the left side of the photo, but in the right side of the photo begins the exclosure, a space that is completely isolated by fencing and the bison cannot enter. Notice the taller vegetation in the exclosure. Numerous exclosures are found throughout the bison unit.  The author (L.D. Fannin) in his mobile lab processing collected grass plants in the field at Nachusa in June 2021. To prevent molding, grass plants need to be dehydrated before transport, and the food dehydrator at the far right allows for the dehydration of plant remains to occur quickly before plants are then ground and stored with desiccant. The author is currently analyzing these collected plant parts for their silica content at Dartmouth.
Luke Fannin was a 2021 Scientific Research Grant recipient from the Friends of Nachusa Grasslands. Interested in supporting Nachusa's science? Just designate your Donation to "Scientific Research Grants."
By Elizabeth Bach Research Scientist at Nachusa Grasslands, The Nature Conservancy Nachusa has experienced several ups and downs in 2021. COVID-19 continued to bring challenges and require flexibility. We celebrated the opening of our new equipment barn, but we also grieved the loss of naturalist Wayne Schennum. Wayne conducted plant and insect surveys at Nachusa throughout his long career and was actively working on a survey of leaf beetles prior to his passing this summer. Amid this uncertainty, the Nachusa science community managed to accomplish a lot.
Pete Guiden started 2021 strong with the publication of Effects of management outweigh effects of plant diversity on restored animal communities in tallgrass prairie in the Proceedings of the National Academy of Sciences. This is one of the top three scientific journals in the world; it is a major accomplishment for Pete and his co-authors in the Holly Jones and Nick Barber labs. It is significant for a Nachusa dataset to contribute to global scientific advancement in this way. They found management-driven responses were more common (and stronger) than plant-driven responses. Restoration age was the main driver of management effects, followed by prescribed fire. Both plant diversity and active management were critical to restoring animal biodiversity. You can learn more from Pete’s blog post. Another significant Nachusa publication was Twenty years of tallgrass prairie restoration in northern Illinois, USA, published in Ecological Solutions and Evidence as part of a global special issue on the UN Decade on Restoration. Elizabeth Bach analyzed 20 years of plant survey data from permanent transects set up by Bill Kleiman. Plant communities on native prairie remnants have maintained or increased plant diversity, including rare plants. Savannas maintained similar levels of plant diversity, but plant communities shifted from understories dominated by brush to herbaceous plants, including native grasses and flowering plants. The special issue on the UN Decade on Restoration also featured a paper from Bethanne Bruninga-Socolar and Sean Griffin. Variation in prescribed fire and bison grazing supports multiple bee nesting groups in tallgrass prairie showed that a mixture of fire and grazing on the landscape encouraged diverse bee communities by promoting bees with different nesting habits. Nachusa bees were primarily ground-nesting (90% of observed species), but stem/hole nesters (3.6%) and large-cavity nesters (6%) are also important parts of the community. This team also published Bee communities in restored prairies are structured by landscape and management, not local floral resources in Basic & Applied Ecology. Large-landscape restorations were positive for bees, as the relationships with floral diversity observed in previous work in small, isolated prairies did not hold up at Nachusa. Several Nachusa insect studies were published in 2021. Michele Rehbein, who surveyed mosquitoes at Nachusa for her PhD work at Western Illinois University, published A new record of Uranotaenia sapphirina and Aedes japonicus in Lee and Ogle Counties, Illinois. Both these mosquitoes are new records for the area and contribute to broader understandings of mosquito communities in Illinois generally. Azeem Rhaman published Disturbance-induced trophic Niche shifts in ground beetles (Coleoptera: Carabidae) in restored grasslands, summarizing his MS research with Nick Barber at San Diego State University. He found ground beetles consumed a wider range of food sources in areas with bison grazing, particularly with both grazing and fire. Meghan Garfinkel, who earned her PhD from University of Illinois – Chicago, specifically examined insects present in bird diets. Using faecal metabarcoding to examine consumption of crop pests and beneficial arthropods in communities of generalist avian insectivores, published in Ibis, was the first study to leverage next-generation DNA sequencing to analyze diets of entire bird communities. Birds consumed more herbivorous arthropods (plant-eating bugs) compared to carnivorous arthropods (bug-eating bugs). Heather Herakovich published two papers on birds this year. In Impacts of a Recent Bison Reintroduction on Grassland Bird Nests and Potential Mechanisms for These Effects, she found that bison presence did not impact nesting density or vegetation structure, but nest success increased in the first two years after reintroduction. Heather evaluated birdsong to passively survey communities in Assessing the Impacts of Prescribed Fire and Bison Disturbance on Birds Using Bioacustic Recorders. This dataset showed that having a mix of recently-burned, unburned, and grazed habitat in the landscape supported diverse bird communities, as different species have different habitat preferences. It was a strong year for animal publications from Nachusa. Rich King and graduate students Monika Kastle and Callie Golba published two papers about their work on Blanding’s turtle recovery in northern Illinois, including the Nachusa population. Blanding's Turtle Demography and Population Viability focused on modeling needs for Blanding’s turtle populations to sustain themselves. Blanding's Turtle Hatchling Survival and Movements following Natural vs. Artificial Incubation reported on the results from the 2020 release of Blanding’s head-start hatchlings at Nachusa and other sites. Survival rates were variable across the populations, and research continues at Nachusa to find the most effective ways to protect these turtles. Publications of threatened and endangered species extended to plants as well. Katie Wenzell, who earned her PhD from Northwestern/Chicago Botanic Gardens, published Incomplete reproductive isolation and low genetic differentiation despite floral divergence across varying geographic scales in Castilleja in the American Journal of Botany. This work examined the genetic relatedness and floral variability of Castelleja sessiliflora (downy paintbrush) and C. purpurea (prairie paintbrush or purple paintbrush) across their geographic ranges. Both species are evolving, but C. sessiliflora is exhibiting genetic differentiation, whereas C. purpurea is exhibiting differences in flower shape without genetic changes. These are two different mechanisms driving similar evolutionary outcomes. Timothy Bell and colleagues explored population trends in the threatened eastern prairie fringed orchid in Environmental and Management Effects on Demographic Processes in the U.S. Threatened Platanthera leucophaea (Nutt.) Lindl. (Orchidaceae). They found regular burning and wet weather lead to greater blooming populations for the orchid. To evaluate landscape-level plant community and soil characteristics, Ryan Blackburn tested aerial imaging from drones in Monitoring ecological characteristics of a tallgrass prairie using an unmanned aerial vehicle, published in Restoration Ecology. Drone images did an adequate job of evaluating grass cover and mean dead plant cover, but more work is needed to refine this method. Scientific accomplishments at Nachusa are making strong contributions to both scientific understanding and on-the-ground conservation and restoration efforts. Thank you all for being part of this. None of this could happen without the collaboration of scientists, volunteers, donors, The Nature Conservancy, and Friends of Nachusa Grasslands. Many of the 2021 researchers were supported with a Scientific Research Grant from the Friends of Nachusa Grasslands. Donations to Friends can be designated to Scientific Research Grants.
The Nachusa summer science externship is supported by The Nature Conservancy. |
Blog CoordinatorDee Hudson
I am a nature photographer, a freelance graphic designer, and steward at Nachusa's Thelma Carpenter Prairie. I have taken photos for Nachusa since 2012. EditorJames Higby
I have been a high school French teacher, registered piano technician, and librarian. In retirement I am a volunteer historian at Lee County Historical and Genealogical Society. Categories
All
Archives
January 2024
|
CONNECT WITH US |
|






























































































































 RSS Feed
RSS Feed